Search Thermo Fisher Scientific
Invitrogen
Mogamulizumab Chimeric Recombinant Rabbit Monoclonal Antibody (KW-0761 (Mogamulizumab))
图: 1 / 1
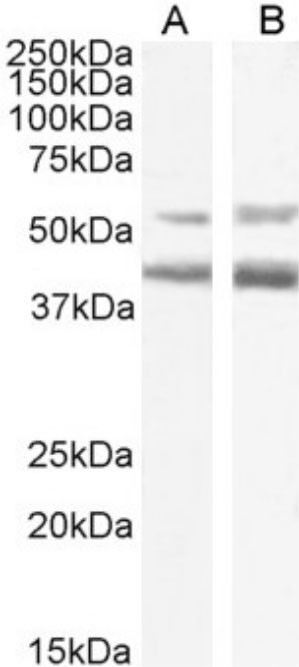
Mogamulizumab Chimeric Antibody (MA5-48050) in WB
产品信息
MA5-48050
种属反应
宿主/亚型
Expression System
分类
类型
克隆号
抗原
偶联物
形式
浓度
纯化类型
保存液
内含物
保存条件
运输条件
RRID
产品详细信息
Specificity: This antibody is specific for human CCR4, a chemokine receptor that is preferentially expressed by Th2 and regulatory T (Treg) cells. CCR4 is also detected on basophils, platelets, monocytes and brain microvascular and coronary artery endothelial cells.
靶标信息
Mogamulizumab is a humanized monoclonal antibody (mAb) directed against CC chemokine receptor 4 (CCR4) for the treatment of Mycosis Fungoides (MF) and Sezary Syndrome (SS), the most common subtypes of cutaneous T-cell lymphoma. Cutaneous T-cell lymphomas occur when certain white blood cells, called T cells, become cancerous; these cancers typically affect the skin, causing various types of skin lesions 8. On August 8 2018, the U. S. Food and pharmaceutical Administration (FDA) approved mogamulizumab injection (also known as Poteligeo) for intravenous use for the treatment of adult patients with relapsed or refractory mycosis fungoides (MF) or Sezary syndrome (SS) after at least one prior systemic therapy 7. Mogamulizumab is derived from Kyowa Hakko Kirin's POTELLIGENT (®) technology, which produces antibodies with enhanced antibody-dependent cell-mediated cytotoxicity (ADCC) activity. Approval in Japan was granted on April 30 2012 by the Japanese Ministry of Health, Labor and Welfare for patients with relapsed or refractory CCR4-positive adult T-cell leukemia-lymphoma 2.
仅用于科研。不用于诊断过程。未经明确授权不得转售。
篇参考文献 (0)
生物信息学
蛋白别名: AMG-761; KW-0761